Avogadro NumberAny gas has the same number of molecules per gram of molecular weight. It is known as Avogadro's number. Several techniques have been used to fix its value at 6.02x1023. One of the five universal constants is Avogadro's number. The Roman letter N is used to represent it. 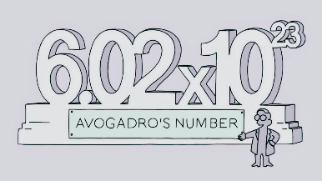
This relationship was formerly known as Avogadro's hypothesis, but after it was tested, it became Avogadro's principle. It is now known as "Avogadro's law." This rule has been utilised to modify atomic theory and explain Gatluak's rule. This rule is employed in the analysis of gases and in determining the molecular weight, the relationship between the mass and volume of gases, and the atomicity of elemental gases. Importance of Avogadro's ConstantSimply expressed, it links the amount of substance to the number of particles, bridging the gap between the large and microscopic worlds. It additionally shows how bound physical constants and characteristics relate to one another. Among them are:
In chemistry, we tend to continuously live things on a large scale. We tend to live things sort of a substance's mass, temperature, or total volume additionally to different general quantities. However, if we tend to examine this from an Associate in Nursing atomic perspective, understanding particle momentum and rate is crucial. Mass is critical. Each of them area unit connected by a constant. How was the Avogadro Number calculated?Avogadro was a baby throughout an important time in the history of chemistry. Chemists like a chemist and Joseph Louis physicist mentioned , however these infinitesimally little particles behaved as they began to comprehend the elemental characteristics of atoms and molecules. Avogadro was notably intrigued by Gay-law Loussac's blending volumes. Avogadro competes with the implications of this law and expects that for it to carry true, equivalent volumes of any 2 gases at comparable weights and temperatures should hold the same variety of particles. And therefore, the sole thanks to prove that this law may well be correct was if there has been a distinction between atoms and molecules and if bound components, like atomic number 7, genuinely existed as molecules (N2 instead of merely N). on condition that Amedeo Avogadro lacked word like "molecule" to explain his hypothesis which his concepts encountered resistance from folks like a chemist, among others, another chemist, Stanislao Cannizzaro, was needed to grant Avogadro's theories the eye they due. Amedeo Avogadro had already passed on to the great beyond by the time their ideas had nonheritable quality. The number was given Avogadro's law's honor by individual Jean Baptiste Perrin since it had been, therefore, vital to the event of chemistry Significance of Avogadro's NumberA substance's mass is measured at the atomic level mistreatment Associate in Nursing mass unit. The definition of an Associate in Nursing mass unit is that the weight of 1 atom is divided by twelve. For instance, the atomic number has an Associate in Nursing mass unit of 1.00794 AMU. It's not possible to see Associate in Nursing atoms, electrons, or molecules' capability to, say, do a reaction.Chemists instead devised a way of connecting the mass unit and also the gram.
We can convert between measure in grams and, therefore, the mass unit's invisible unit of measure victimisation. this is often the connexion of Avogadro's number as a result. History of Avogadro's NumberAmedeo physicist, AN Italian man of science, is worthy by having his variety named once him. The physicist didn't counsel the constant, however, he did postulate that the quantity of a gas at a selected temperature and pressure was proportionate to the number of particles it contained. French man of science Jean Perrin constantly instructed in 1909. He applied a variety of techniques to work out the constant's price, earning him the 1926 honour in Physics. Perrin's price, however, supported the number of atoms contained in a single gram-molecule of an atomic, chemical element. A later definition of the constant used twelve grams of carbon-12 because of the basis. The amount is additionally observed because of the Loschmidt constant in German literature. How Big Is Avogadro's Number?This is primarily identical as asking however huge a mole is as a result of Avogadro's number equals one mole. Avogadro's number is applied to anything:
Next TopicBar Graphs
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










