Dialysis DefinitionWhen a person's kidneys cannot work normally, kidney dialysis removes extra water, solutes, and toxins from the blood. The term "dialysis" comes from the Greek word "dialysis," which means "dissolution"; "dia" means "through," and "lysis" means "loosening or splitting." Renal replacement therapy is the term used to describe this treatment. In 1943, the first successful dialysis procedure was carried out. Acute kidney damage, also known as acute renal failure, is a quick, abrupt loss of kidney function that may need the start of dialysis. A progressive decrease of kidney function is a chronic renal disease, sometimes called stage 5 kidney disease. When the creatinine clearance falls below 10 mL per minute, uremia is present, and the glomerular filtration rate is between 10% and 15% of normal, stage 5 chronic renal failure is reached. 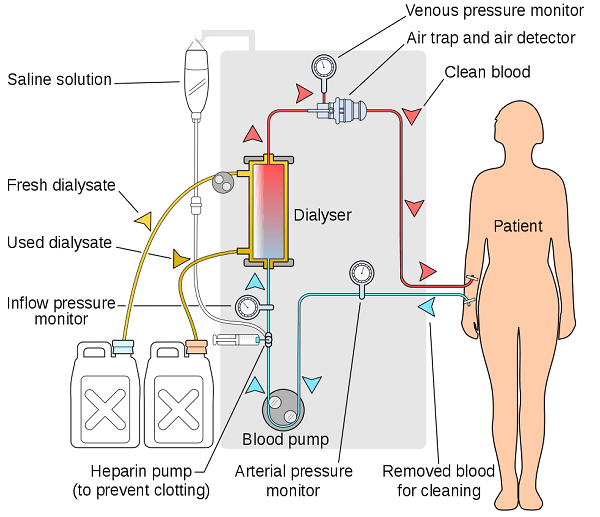
Dialysis is a temporary solution when someone has an acute renal injury or is waiting for a kidney transplant. It is also used as a permanent solution when a transplant is neither recommended nor practicable. In Australia, Canada, the UK, and the US, the government covers the cost of dialysis for those who qualify. In research labs, the dialysis procedure can segregate molecules based on size. Using this technique, the buffer (sometimes referred to as "dialysis bath" or "dialysate") between a sample and the solution can also be balanced. A tubular semipermeable nitrocellulose or cellulose acetate membrane is used for dialysis in a lab setting. 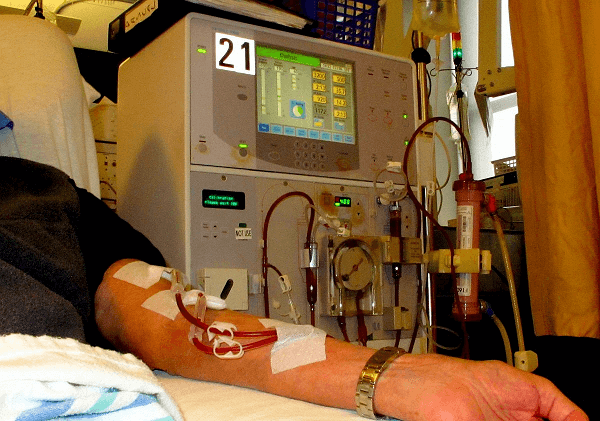
The needed size separation determines the pore size, with bigger hole sizes allowing larger molecules to flow through the membrane. Larger molecules cannot flow through the semipermeable membrane's gaps, although solvents, ions, and buffers can easily diffuse through it. This can be used to separate larger proteins and molecules from a complicated mixture to purify the desired proteins. In preserving health, the kidneys play a crucial function. The kidneys keep the body's internal mineral and water balance in good condition (sodium, potassium, chloride, calcium, phosphorus, magnesium, sulfate). Also eliminated through the kidneys are the acidic metabolic waste products that the body cannot expel through breathing. Along with manufacturing erythropoietin, calcitriol, and renin, the kidneys perform other endocrine system duties. Red blood cell production is influenced by erythropoietin, and bone development is influenced by calcitriol. Because dialysis does not restore the kidney's damaged endocrine functions, it is a poor treatment to replace renal function. By ultrafiltration and diffusion (waste removal), dialysis treatments replace some of these duties (fluid removal). Highly purified (sometimes called "ultrapure") water is used during dialysis. Principle:In dialysis, a semipermeable membrane filters fluid and disperses solutes over it. The diffusion property, which is the inclination for chemicals to travel from a location of high concentration to a low concentration, is demonstrated by chemicals in the water. A semipermeable membrane has two sides: one is used to pass blood, while the other is used to pass dialysate, a particular dialysis fluid. A thin layer of material with pores of different sizes is called a semipermeable membrane. Fluids and smaller solutes can pass across the membrane, but larger things cannot (for example, red blood cells and large proteins). The glomerulus replicates the cleaning process in the kidneys by separating the larger chemicals from the smaller ones as blood enters the kidneys. The two main dialysis procedures?hemodialysis and peritoneal dialysis?each have a different way of removing waste materials and excess water from the blood. By pumping blood outside the body via a semipermeable membrane-containing external filter known as a dialyzer, hemodialysis eliminates wastes and water from the blood. The dialysate flows in the opposite direction from the blood, which flows in one direction. More urea and creatinine are removed from the circulation due to the blood and dialysate flowing in opposite directions. This improves the gradient of concentration of solutes between the dialysate and blood. The dialysate must be replaced frequently to maintain a low concentration of unwanted solutes on this side of the membrane. It is undesirable for solute concentrations generally found in urine (potassium, phosphorus, and urea) to be high in blood but low or missing in the dialysis solution. Mineral concentrations in the dialysis fluid, such as potassium and calcium, are comparable to those in normal blood. The sodium content in the dialysate is set slightly higher than that in normal blood to aid in sodium absorption into the blood and act as a ph to correct the alkalosis that is generally present in these patients. Typically, a nephrologist will prescribe the levels of the dialysate's components based on the specific patient's requirements. For peritoneal dialysis, which removes wastes and water from the circulation inside the body, the peritoneum is used as a natural semipermeable membrane. A specific dialysis solution called dialysate is created in the abdominal cavity by moving wastes and extra water out of the circulation and over the peritoneal membrane. Types:Hemodialysis, peritoneal dialysis, hemofiltration, hemodiafiltration, and intestinal dialysis are the four major and secondary forms of dialysis, respectively (secondary). 1. Hemodialysis:The patient's blood is exposed to a partially permeable membrane during hemodialysis by being pushed via the blood compartment of a dialyzer. Thousands of microscopic hollow synthetic fibers are used to make the dialyzer. The membrane is a semipermeable structure made of fibers. Water and wastes are transported between the dialysis solution and blood as they move around the fibers' exterior. After that, the blood is circulated back to the body after being cleaned. Higher hydrostatic pressure is applied across the dialyzer membrane to create ultrafiltration. For this purpose, negative pressure is frequently applied to the dialysate compartment of the dialyzer. Because of the pressure gradient that causes water and other dissolved solutes to move from blood to dialysate throughout a standard 4-hour therapy, additional fluid can be eliminated. Hemodialysis in the United States is normally performed three times a week in a dialysis facility (because of Medicare reimbursement rules in the United States), but, as of 2005, over 2,500 Americans were dialyzing at home more regularly for varying treatment durations. Dialyzing for 6 to 8 hours, 5 to 7 times per week, has been shown to have positive clinical effects in studies. It has been demonstrated in a study that this type of hemodialysis, also known as nocturnal daily hemodialysis, significantly improves both small and big molecular weight clearance and reduces the requirement for phosphate binders. Home dialysis can change schedules daily or weekly, making it a flexible treatment choice. These frequent, lengthy treatments are frequently performed at home while you are asleep. Most research supports the therapeutic advantages of longer treatment times and more frequent appointments. In 2011, 909,000 hospital stays included hemodialysis, making it one of the most frequent procedures in US hospitals (a rate of 29 stays per 10,000 population). 2. Peritoneal dialysis:In peritoneal dialysis, the abdominal cavity surrounding the intestine is filled with a sterile solution containing glucose (dialysate) injected through a tube. The peritoneal membrane functions as a partially permeable membrane in this situation. This exchange happens four to five times daily; automated systems can do more frequent exchange cycles at night. Although peritoneal dialysis is less effective than hemodialysis, it produces outcomes comparable to hemodialysis in waste product elimination and salt and water removal since it is performed longer. In many cases, the patient performs peritoneal dialysis by themselves at home. Patients are thus released from the monotony of having to visit a dialysis facility regularly, numerous times each week. Peritoneal dialysis can be carried out with little to no specialized equipment. 3. Hemofiltration:While hemodialysis and hemofiltration are similar treatments, they operate on different principles. Instead of using dialysate, the blood is pushed through a dialyzer or "hemofilter" like in dialysis. The permeable membrane experiences a pressure gradient, which causes water to travel through it quickly. As it does so, it "drags" along numerous dissolved chemicals, including some with large molecular weights that hemodialysis cannot remove as effectively. The extracorporeal circuit is continuously fed with a "substitution fluid" to replenish salts & water lost from the blood during the treatment. 4. Hemodiafiltration:When the kidneys are not functioning correctly, hemodiafiltration, a mix of hemodialysis and hemofiltration, removes toxins from the blood. It is also used to treat acute renal injury (AKI). Dialysis of the Intestines:In intestinal dialysis, soluble fibers are added to the food as a supplement, such as acacia fiber, broken down by colon bacteria. The nitrogen level excreted in fecal waste rises due to this bacterial development. Ingestion of 1 to 1.5 liters of non-absorbable polyethylene glycol or mannitol solutions every four hours is an alternative strategy. Indications:Patients with renal failure must consider several considerations before starting either hemofiltration or dialysis. Both acute and chronic indications can be used to categorize them. There may be an overlap between the symptoms of depression and kidney failure. A dialysis team and the patient must be able to communicate openly. Giving improved living quality is possible with open communication. The dialysis team can offer more options if they know the patient's demands. For example, they can adjust the type of dialysis so that patients can be more active at home, or they might change 3the patients' eating habits so that they don't produce unneeded waste. 1. Acute indications:Using the vowel mnemonic "AEIOU," the following are the indications for dialysis in a patient with acute kidney injury:
2. Chronic indications:Chronic dialysis may be required when a patient has a symptomatic renal impairment and a low glomerular filtration rate (GFR 15 mL/min). The estimated GFR, or eGFR, at which dialysis started increased between 1996 and 2008. Early dialysis commencement, defined as starting dialysis with an estimated GFR of more than 10 ml/min/1.732, does not appear to have any benefits and may even have negative effects. Early initiation to dialysis may be hazardous, according to observational evidence from sizable registries of dialysis patients. According to Canada's most recently published guidelines, patients should wait to start dialysis until they have clear signs of renal failure, which may start to show up at a GFR estimate of 5 to 9 ml/min/1.732. Pediatric Dialysis:Children have benefited greatly from significant technological advancements and clinical dialysis management during the past 20 years. Seizures are now unusual during dialysis sessions, and hypotensive events are now uncommon. Chronic internal jugular venous catheters and anesthetic creams for fistula punctures have helped lessen pain and suffering. Patient morbidity and medical expenses can greatly decrease using non-invasive devices to measure patient target dry weight and access flow. Mortality in children and young adults receiving chronic hemodialysis is linked to several factors, including nutrition, inflammation, anemia, and the amount of dialysis given. This emphasizes the need for multimodal intervention strategies in addition to adequate hemodialysis treatment as determined by Kt/V alone. 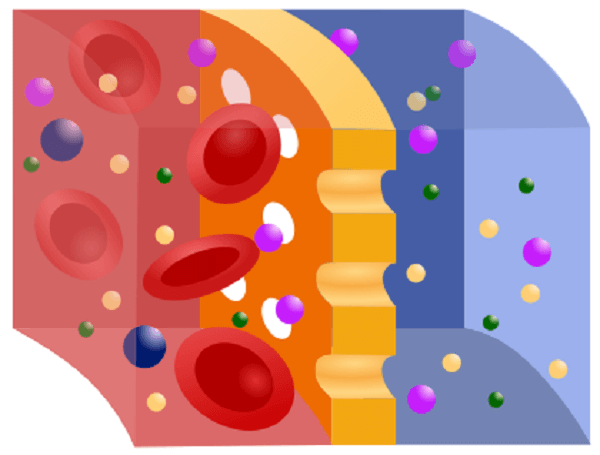
New low extracorporeal volume tubing, biocompatible synthetic membranes, and small-size dialyzers have been created for young newborns. Regardless of the blood pump segment size, which can range from 6.4 mm for regular dialysis to 8.0 mm for high flux dialysis in all patients, the arterial and venous tubing length is constructed of a minimum length and diameter. A tubing with a volume of between 80 and 110 ml is designed for pediatric patients, and a tubing with a volume of between 130 and 224 ml is designed for adult patients. Each dialysis machine manufacturer builds their system with pediatric dialysis in mind. The pump speed should be slowed in pediatric patients by the patient's blood production capacity, and the heparin dose should be regularly monitored for clotting. Patients under the age of 18 shouldn't undergo high-flux dialysis. 
Hemodialysis in children must be tailored to each patient's specific needs and considered an "integrated therapy" considering the patient's prolonged exposure to chronic renal failure therapy. Compared to renal transplantation, dialysis is only viewed as a temporary solution for children because it offers the highest possibility of rehabilitation in terms of educational and psychological functioning. Yet to protect their future "cardiovascular life," the greatest criteria should be applied to the long-term chronic dialysis of young kids. Longer dialysis sessions, online hemodiafiltration using synthetic high flux membranes with surfaces ranging from 0.2 m2 to 0.8 m2, and blood tubing lines with a low volume yet large blood pump section may be necessary to expand on the very limited notion of small-solute urea dialysis. Several Nations' Experiences with Dialysis:In the UK:In the UK, dialysis is offered by the National Health Service. NHS England in England is the organization that commissions the service. The program is utilized by about 23,000 patients annually. Patient transportation services are typically offered free of charge for patients who need to travel to dialysis facilities. Following a campaign started by Kidney Care UK, the Cornwall Clinical Commissioning Group decided to fund transport services for patients who required dialysis at least six times per month for at least three months or at least three times per week for at least three months. Originally, they had proposed restricting this provision to patients without specific medical or financial needs. According to a UK study, dialysis at home is less expensive than dialysis in a hospital. Yet, many people in the UK choose to do dialysis in a hospital for various reasons, including daily social interaction. To save money for the NHS and lessen the negative effects of dialysis on people's social and professional lives, the NHS might encourage patients to get their dialysis at home. In the USA:All Americans' insurance premiums in the US have been paid for transplants and dialysis since 1972. By 2014, over 460,000 individuals were receiving treatment, with the associated expenses making almost 6% of the entire Medicare budget. The United States has one of the highest mortality rates for dialysis care in the industrialized world, and kidney disease is the ninth greatest cause of death worldwide. Patients are receiving kidney transplants at a lower rate than anticipated. A new for-profit dialysis industry that emerged in response to changes in government payment policy has been blamed for these results. Patients treated in for-profit dialysis centers had greater mortality rates & are less likely to be included on the kidney transplant waiting list than patients treated in non-profit institutions, claims a 1999 study. This may be because a kidney transplant eliminates the facility's ability to generate a steady income. The insurance sector has voiced concerns about kickbacks and unsatisfactory connections between charity and service providers. In China:The Chinese government financially supports dialysis treatment. Due to the uneven distribution of dialysis facilities and healthcare resources, reaching everyone who needs dialysis therapy is difficult. In China, hemodialysis or peritoneal dialysis is administered to 395,121 patients annually. Chronic renal disease affects 10.8% of the population in China. To address the demands of those suffering from chronic renal disease in the country, the Chinese government is attempting to increase the volume of peritoneal dialysis. History:The first dialysis machine was created in 1913 by Johns Hopkins Hospital employees Leonard Rowntree and John Abel. They tried it on animals with great success. In 1943, when the Netherlands was under German control, a Dutch physician named Willem Johan Kolff built the first functional dialyzer. Kolff had to improvise and construct the first machine utilizing materials that were readily available at the time, including a washing machine, soda cans, and sausage casings, due to the lack of resources. Kolff attempted to use his device to treat 16 patients with acute kidney failure over the next two years (1944-1955), but the treatments failed. Then, in 1945, a 67-year-old woman who had been in a coma for 11 hours had hemodialysis using a dialyzer and regained consciousness afterward; she survived for another seven years before passing away from an unrelated illness. She was the very first patient to receive effective dialysis care. A dialysis device was independently created in 1945 by Gordon Murray of the University of Toronto. Murray's machine used fixed flat plates, which was more in line with contemporary designs than Kolff's rotating drum. Like Kolff, Murray also saw early results with individuals with acute renal failure. Nils Alwall of Lund University improved the Kolff dialysis machine in Sweden by enclosing it in a stainless steel canister. The first hemodialysis device was useful since it enabled removing fluids by exerting a negative pressure on the outside canister. On September 3, 1946, Alwall treated his first patient experiencing acute kidney failure. The Conclusion:The necessity to deliver more personalized or personalized care in place of a "one-size-fits-all" approach to dialysis was one of the conference's main themes. It is now acknowledged that setting and achieving patient-centered goals is a crucial aspect of dialysis care. To do this, it will be necessary to use tools for both patients and providers and to align the goals and incentives of patients, carers, healthcare professionals, regulators, and payers. Participants in the meeting acknowledge that any recommendation for shifting to patient-centered, goal-directed dialysis presupposes that any systems now in place for providing dialysis may be changed. Granted, the ability to alter current structures may be constrained in some parts of the world.
Next TopicDigestive System Definition
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










