What is the Full Form of HPLCHPLC: High-Performance Liquid ChromatographyHPLC stands for high-performance liquid chromatography. High-performance liquid chromatography (HPLC) is an analytical chemistry method for separating, identifying, and quantifying each component in a mixture. Previously, it was known as high-pressure liquid chromatography. The sample combination is passed through a column loaded with a solid adsorbent material using pumps to move a pressured liquid solvent through the column. Each component in the sample behaves a bit differently with the adsorbent materials, leading to variable flow rates for the various components and causing the elements to segregate as they exit the column. 
HPLC has been mainly employed in manufacturing (such as during the manufacturing of biological and pharmaceutical goods), legal (such as detecting performance-enhancing drugs in urine), and research (such as separating the constituents of a sophisticated biological sample or identical synthetic chemicals from one another), and medical (such as determining the vitamin D levels in blood serum) purposes. Chromatography can be regarded as an adsorption-based mass transfer procedure. In HPLC, the sample components are separated by pumping a pressurised liquid and a sample combination through a column loaded with adsorbent. The column's adsorbent, or active component, is generally a granular substance comprised of solid particles ranging in size from 2 to 50 m, such as silica or polymers. The varying levels of interaction between the components of the sample mixture and the adsorbent particles cause them to be segregated from one another. The pressurised liquid, also known as the "mobile phase", is often a blend of solvents (such as water, acetonitrile, and methanol). The interactions between the components of the sample and the adsorbent are influenced by its composition and temperature, which are important factors in the separation process. These interactions, which are typically combinations, are physical by nature, such as hydrophobic (dispersive), dipole-dipole, and ionic. Operational pressures are substantially higher (50-350 bar) in HPLC than in conventional ("low pressure") liquid chromatography. In contrast, in conventional liquid chromatography, the mobile phase is normally passed through the column by the gravity force. The average column dimensions for analytical HPLC are 2.1 - 4.6mm in diameter and 30 - 250mm in length due to the limited sample amount that is separated. Additionally, HPLC columns are created using tiny adsorbent particles (2 - 50m in average particle size). This has made HPLC a widely known chromatographic method, giving it higher resolving power (the capacity to differentiate between substances) when distinguishing mixtures. A degasser, injector, pumps, columns, column oven and detector, are frequently shown on an HPLC device's diagram. The sampler introduces the sample combination into the mobile phase stream and then transports it to the column. The pumps push the mobile phase through the column at the correct flow and composition. It is possible to analyse the sample components quantitatively since the detector produces a signal proportional to the number of sample components coming from the column. 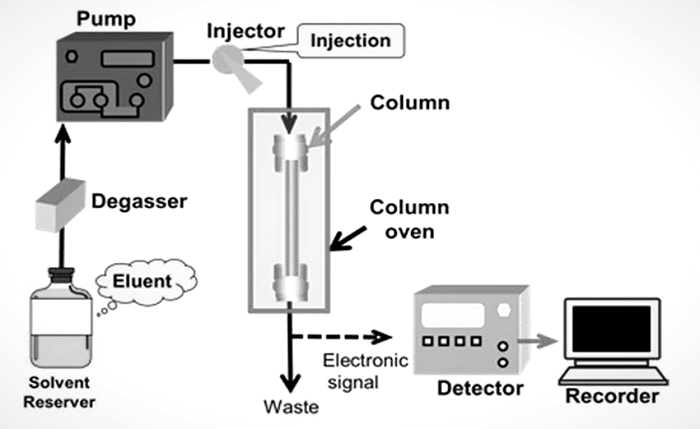
The HPLC apparatus is controlled by a digital microprocessor, and user software offers data analysis. In some HPLC equipment models, the mechanical pumps can combine various solvents in ratios that change over time, creating a composition gradient in the mobile phase. Numerous detectors are frequently used, including UV/Vis, photodiode arrays (PDA), and mass spectrometry-based detectors. The majority of HPLC devices additionally contain a column oven that enables temperature adjustment during separation. TypesOther than high-performance liquid chromatography, there are mainly the following types of chromatography:
ApplicationsManufacturingHPLC has several uses in the laboratory and in the field of clinical science. Since it is a reliable method for obtaining and ensuring product purity, it's a common approach employed in creating pharmaceuticals. Even though HPLC can create products of incredibly high quality (purity), it isn't always the main technique employed in manufacturing bulk medicinal ingredients. HPLC improves specificity, precision, and accuracy, but regrettably, this also results in a rise in cost. Given that HPLC on a big scale could be an expensive technology, this, however, might be the result of different time and financial restrictions. LegalAnother use of this technology is to identify illicit drugs in urine. An immunoassay is the most popular method of drug detection. This approach is considerably more feasible. But convenience comes at the expense of clarity and comprehensive drug coverage. Using HPLC alone to assess drug concentrations is inadequate because it is a technique for determining (and maybe increasing) purity. Due to this, mass spectrometry and HPLC are frequently used in this situation. The absolute requirement for standardising HPLC experimental runs is reduced when mass spectrometry is used in conjunction with HPLC. ResearchResearch can use similar techniques to find potential drug concentrations in antifungal and asthma drug candidates. Since purity is crucial in this kind of research, it is utilised to verify the outcomes of synthesis reactions. Mass spectrometry, however, continues to be a more trustworthy method for identifying unknown compounds within a sample. This method works well for observing several compounds in samples that have been gathered, but it is necessary to utilise standard solutions when trying to identify the compounds.
Next TopicFull Form
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










