Oxidation and ReductionOxidation often occurs all around us. For example, a sliced apple turns brown after sometime and peeled potatoes also change their colour after sometime. These changes occur due to oxidation. Similarly, reduction processes also occur in our daily life such as photosynthesis in which carbon dioxide undergoes reduction and converted to carbohydrate. What is oxidation?It is a chemical process that can be understood or explained in four different ways as described below;
So, oxidation is a chemical process in which any of the above four processes can take place. For example; i) Transfer of electrons: The transfer of electrons takes place in the oxidation of chemical species like atoms, elements or molecules. For example, in the below chemical reaction, we can observe the loss of electrons or transfer of electrons. The oxidation of magnesium occurs as it loses electrons which are gained by oxygen to form Magnesium oxide. 2 Mg (s) + O2 → 2 MgO (s) In the above reaction, Mg is losing two electrons to form Mg2+ and oxygen gains 2 electrons to form O2+. The Mg2+ combines with O2- to form MgO molecules in which opposite charges of oxygen and magnesium ions neutralize each other to form a neutral molecule. In this case, Mg is losing electrons, which is actually oxidation. Whereas, oxygen gains electrons, which is actually a reduction. So, if an element is gaining electrons, its reduction occurs. ii) Change in oxidation number: The oxidation state or number increases if oxidation occurs. For example, the oxidation number of magnesium increases from 0 to +2. As one atom of magnesium loses two electrons. Similarly, when sodium reacts with chlorine to form sodium chloride. The oxidation state or number of sodium increases from 0 to +1. iii) Transfer of oxygen: The transfer of oxygen or the addition of oxygen to a substance also shows oxidation. See the below reaction; For example, 2Mg + O2 → 2MgO In the beginning, the magnesium was alone and after reacting with oxygen, oxygen is added to it magnesium and thus oxidation of magnesium takes place. So, the reaction in which oxygen is added to a substance or reactant is called an oxidation reaction. iv) Transfer of hydrogen: The hydrogen is lost by the reactant which is oxidised. For example, in the blow reaction, ammonia is oxidised by losing hydrogen to bromine. 2NH3 + 3Br2 → N2 + 6HBr So, the removal of hydrogen from a substance is also known as oxidation of that substance. Similarly, in the below reaction; NH3 → N2 + H2 Nitrogen is with hydrogen (NH3) before the decomposition of ammonia. After the decomposition hydrogen is separated from nitrogen to form N2 and H2. So, in this case, ammonia is oxidised, so it is an oxidation reaction. There is no reaction in which only oxidation or reduction occurs. So, oxidation and reduction occur simultaneously and such chemical reactions in which they occur simultaneously are called redox reactions. For example; in the below reaction in which Zn reacts with copper sulphate to form zinc sulphate and copper is a redox reaction. Zn + CuSO4 (aq) → ZnSO4 (aq) + Cu In the beginning, Zn was alone but after the reaction, it comes with oxygen in Zinc sulphate (ZnSO4) and the Cu leaves the oxygen. We can say that Zn is adding oxygen to it so it is oxidising itself. Now, CuSO4 is converted to Cu so copper sulphate is undergoing reduction. Zn is losing two electrons and becomes Zn2+ so it has been oxidized in this reaction. Cu2+ is converted to Cu by gaining two electrons so it undergoes reduction. So, it is a redox reaction as both oxidation and reduction occur simultaneously. Similarly, the below reaction is also a redox reaction, as both reduction and oxidation are taking place as shown in the below image. Iron oxide is undergoing reduction to produce iron, whereas, carbon monoxide undergoes oxidation to form carbon dioxide. 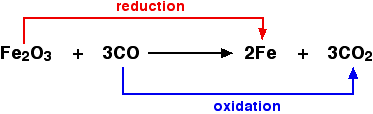
Examples of OxidationOxidation occurs everywhere around us. The common examples of oxidation are as follows; 1. Rusting Oxidation causes changes in the properties of chemical species that undergo oxidation. For example, rusting of iron when it reacts with oxygen. Iron is a solid metal before oxidation but after oxidation, it becomes rust which is actually iron oxide. Water is also required in the rusting of iron, it acts as an electrolyte. 2. Fruit The sliced apple pieces turn brown when left open after sometime. This is because when the skin of the apple is removed, the cells are exposed to oxygen and a chemical reaction takes place that involves oxidation due to which apple pieces change their colour. 3. Fire When a substance burns and produces fire then it also involves oxidation. An oxidizing agent is required to cause burning or fire. Oxygen in the air is the most commons oxidizing agent used for burning. Besides these halogens, hydrogen peroxide also acts as a good oxidizing agent. 4. Ethanoic acid Oxidation of alcohol takes place when ethanol is converted into ethanoic acid. Potassium dichromate with dilute sulphuric acid is commonly used for the oxidation of ethanol. In this reaction, the oxidation involves the loss of hydrogen from ethanol, which becomes acetaldehyde. The acetaldehyde is then oxidised into ethanoic acid by gaining an oxygen atom as shown in the below reaction; CH3CH2OH + O → CH3CHO + O →CH3COOH 5. Biological Oxidation of alcohol The oxidation of alcoholic beverages also occurs in our bodies. The ethanol is converted into ethanoic acid in our bodies through oxidation. This oxidation process is catalysed by enzymes present in our body. ReductionWhat is Reduction?Reduction is a chemical process that takes place in all redox reactions like oxidation. Just like oxidation, the reduction can be understood or explained in four different ways as described below one by one. i) Oxygen transfer: Reduction involves loss of oxygen. The atom which loses oxygen is said to be reduced. For example, in the below chemical reaction, the copper in the copper oxide molecule loses oxygen and thus gets reduced to a single copper atom. CuO + Mg → Cu + MgO Similarly, in the below reaction, Mg is with oxygen in the beginning or before the reaction. After the reaction, Mg gets separated from oxygen, and thus MgO is reduced. MgO → Mg + O2 Similarly, copper oxide is reduced to copper by losing oxygen when it reacts with hydrogen gas. The hydrogen gets oxidised to form water as shown below; CuO + H2 → Cu + H2O ii) Electron transfer: Reduction involves the gain of electrons. The atom or element that gains electrons is said to be reduced. For example; In the below reaction, the oxygen atom gains electrons and forms an oxygen ion (O-2). So, the reduction of oxygen takes place as it gains electrons from magnesium atoms. 2Mg (s) + O2 (g) → 2MgO (s) The formation of non-metal ions of metal also involves reduction (gain of electrons) during electrolysis such as; Cl2 +2e- →2Cl- Similarly, positive metal ions are reduced to metals after gaining electrons, for example; Cu2+ + 2e- →Cu iii) Hydrogen transfer: The transfer of hydrogen also indicates that reduction has occurred. The chemical species that gains hydrogen is said to be reduced. For example, see the below reaction; Mg + H2 → MgH2 Mg was alone in the beginning, after reacting with hydrogen gas, hydrogen is added to it and thus magnesium gets reduced. So, the reaction in which hydrogen is added to a substance is called a reduction reaction. Similarly, aldehydes and ketones are reduced to primary and secondary alcohols by adding hydrogen as shown below; CH3CHO + 2H → CH3CH2OH iv) Oxidation number: Reduction also involves a decrease in the oxidation state or number of an atom. We can say that the oxidation number of a chemical species decreases by gaining electrons. For example, in the below reaction, the oxidation number of hydrogen decreases from +1 to 0 as +1 > 0. Zn (s) + 2H+ (aq) → Zn2+ (aq) + H2 (g) Similarly, in the following reaction, the oxidation number of copper (Cu) decreases from +2 to 0. CuO + Mg → Cu + MgO Cu2+ + Mg → Cu + Mg2+ Common example of reductionMetal ores: The metal ores which are oxides undergo reduction to produce respective metals. The iron is extracted from iron ore by this method in which carbon monoxide is used as a reducing agent. See below how it happens; Fe2O3 + 3CO → 2Fe + 3CO2 Difference between oxidation and reduction
Next TopicOxidizing Agent
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










