What is 1 Atomic Mass Unit equal to?Knowing the masses of atoms is extremely important because they are the fundamental building blocks of all matter or substances in the world. An atomic mass unit (AMU) is the basic measure that helps quantify the mass of an atom. This is crucial for comprehending how matter is made and how interactions between different particles affect the resultant matter. The atomic mass unit offers a uniform method of measuring masses that enable researchers to precisely compare and contrast the weights of various atoms. AMU enables the analysis of the chemical and physical characteristics of matter, enabling the creation of novel tools and ground-breaking scientific findings. 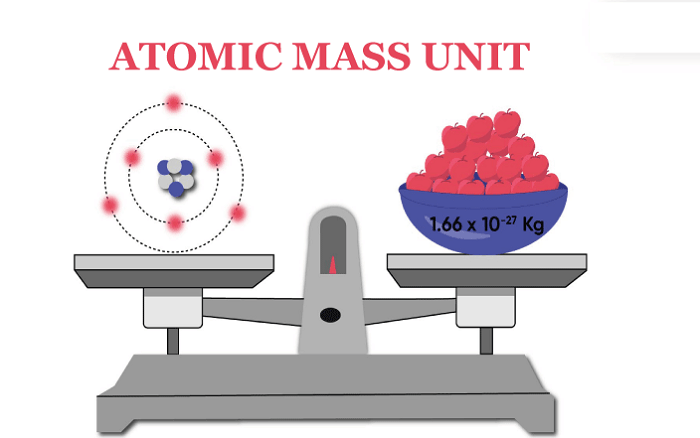
The mass of atoms, compounds, and tiny entities like electrons, protons, and neutrons are all measured using the atomic mass unit. The mass of big assemblages of atoms, like molecules, is also determined using this measure. We will learn in-depth information about the atomic mass unit in this article. Let's start! IntroductionIn 1961, the International Union of Pure and Applied Chemistry (IUPAC) announced the creation of the atomic mass unit. The atomic mass unit was previously known as the Dalton (Da). In 1961, Dalton changed from his initial definition of one-twelfth the mass of a single hydrogen atom to one-twelfth the mass of a single carbon-12 atom. It is also referred to as a unified atomic mass unit, represented by 'u'. As already said, an AMU (or amu) is frequently used to describe the mass of atoms and molecules. For instance, a solitary carbon-12 particle has an atomic mass of 12.011 amu. In other words, 12.011 atomic mass units make up the atomic mass of a single carbon-12 particle. The mass of tiny components like electrons, protons, and nuclei is also expressed using atomic mass units. A proton has a mass of 1.007276 amu compared to an electron's 0.0005486 amu. A neutron has a marginally greater mass of 1.008665 amu approximately. The mass of big assemblages of atoms, like molecules, is also measured using atomic mass units. Water (H2O), for instance, has a molecular mass of 18.015 amu. Accordingly, one water molecule has a mass of 18.015 atomic mass units. So, to conclude, an atomic mass unit is defined as equal to one-twelfth (1/12) of the mass of a carbon-12 atom, which is roughly 1.67377 x 10^ (-24) grams or 1.67377 x is 10^ (-27) Kg. History of Atomic Mass UnitIn science and physics, atomic mass is measured in terms of the atomic mass unit (amu), a measurement of mass. It is specified as being the one-twelfth of the mass of a free carbon-12 (12C) particle when it is at rest. It is occasionally used as a unit of energy because it is a unit of mass. German scientist Wilhelm Ostwald first suggested the AMU in 1883. The mass of an oxygen molecule, which John Dalton calculated in 1803, served as the basis for this unit. Other researchers, including Lord Rayleigh and J.J. Thomson, subsequently improved Dalton's findings. The International Union of Pure and Applied Chemistry (IUPAC) first acknowledged the AMU as a global standard in 1961. The united atomic mass unit (AMU), which has a mass of 1.66053906660(50) x 10^ (-27) kg, is what the IUPAC refers to. This number was calculated using the masses of the carbon-12, oxygen-16, and nitrogen-14 isotopes, which are all naturally present. After that, the IUPAC updated the amu in 2019 to be 1.660538921(73) x 10^ (-27) kg. Based on a more accurate weighted average of the weights of carbon-12, oxygen-16, and nitrogen-14 naturally occurring isotopes, this number was calculated. Uses of Atomic Mass Units in Different FieldsA unit of measurement used to determine the mass of atoms and molecules is the atomic mass unit (AMU). It is almost identical to the energy of a single proton or neutron and equal to one-twelfth of the mass of a carbon-12 particle. In nuclear physics, particle physics, and chemistry, the atomic mass measure is frequently employed. The atomic mass measure is used in chemistry to determine the proportional weights of atoms and molecules. Understanding an atom's chemical and physical characteristics requires being able to measure the masses of various atoms, which is done using the atomic mass unit. For instance, the chemical reactivity of various elements can be determined using their respective weights. The atomic mass unit is used in nuclear physics to calculate the masses of atoms and quantum entities. In order to comprehend nuclear processes like fission and fusion, it is also used to determine the binding energy of atoms. The quantity of energy that can be produced during a nuclear reaction is also measured in terms of the atomic mass unit. The atomic mass unit is used in particle physics to calculate the weights of elementary entities like quarks and leptons. Additionally, it is used to determine the mass of compound elements like mesons and baryons. The mass of electrons created in high-energy collisions, like those at the Large Hadron Collider, is also determined using the atomic mass measure. In general, the atomic mass unit is a significant quantity of measurement used in a number of scientific disciplines. It is used to determine the binding energy of nuclei and the quantity of energy released during nuclear processes. It is also used to measure the masses of atoms, molecules, and particles. Uses of Atomic Mass Units in Different FieldsAlthough it is a useful unit of measurement, there are some drawbacks to using it. The amu is not a basic unit of measurement to start with. It is not an international standard because it is founded on the mass of a particle of carbon-12. Other atoms or compounds may have slightly different masses based on their composition. This makes it challenging to determine an atom's or a molecule's mass with any level of accuracy. Second, changing an amu into another unit of measurement is challenging. Although it is feasible to convert it into other measures, such as kilograms, doing so takes a lot of math and can take some time. Third, AMU's accuracy is subpar. It does not account for the mass of other elements in a molecule and is dependent solely on the mass of a single carbon-12 atom. When determining the mass of molecules, this may result in errors. Uses of Atomic Mass Units in Different FieldsUnderstanding the material world around us requires the use of the atomic mass unit, which is a strong and crucial tool. It has helped us comprehend matter's structure better and given us a foundation on which to learn physics and chemistry. We have made amazing strides in science and technology thanks to its accuracy in measuring atomic mass. For researchers looking to explore and discover, the atomic mass unit will continue to be an invaluable tool.
Next TopicCalculate the pH of 0.05 M NaOH Solution
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










