Gram Positive BacteriaGram-Positive Bacteria are those bacteria that show violet color in the Gram staining test. That is, they show a positive result. Gram staining is a historic procedure commonly used to differentiate the bacteria into two major groups according to the cell wall composition, including thickness. 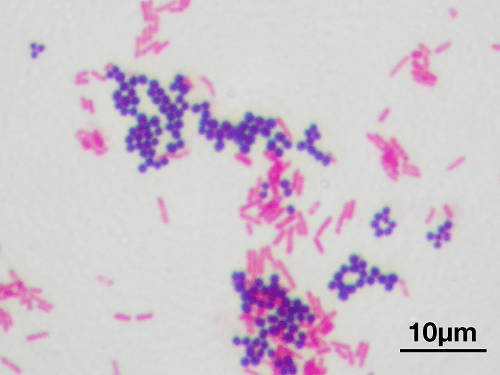
When the bacteria are stained with the dye crystal violet during the staining test, and if the bacteria appear purple under the optical microscope, then they are Grand positive. If they appear pink, then they are Gram-Negative. The reason for this purple color is the Crystal Violet dye is retained after the washing of the sample after the declaration step has been done. This is due to a thick peptidoglycan layer present in the cell wall of Gram-Positive Bacteria. Whereas on the other hand, Gram-Negative bacteria do not have athe alcohothe thick peptidoglycan layer. Hence, they are unable to retain the staining of Crystal Violet dye after decolorization. The dye is retained in the outer membrane of the bacteria. The alcohol employed for decolorization destroys the outer membrane, making the cell wall more porous and resulting in loss of color of the dye. In Gram-Negative bacteria, it is not that the layer is absolutely absent but a thin peptidoglycan layer is present in- between the bacterial outer membrane and the inner cell membrane. It is due to the absence of the outer membrane; Gram-Positive Bacteria are thought to be more susceptible to the antibiotics that target the cell wall of bacteria. On the contrary Gram-Negative bacteria possess a thick outer membrane and are resistant to these antibiotics. This thin layer can only absorb the counterstain, which is saffron and or fuschin, and therefore it appears red or pink under the optical microscope. Apart from the thickness of the peptidoglycan layer, another difference between Gram-Negative and Gram-Positive Bacteria is the presence of the outer membrane, which is absent in Gram-positive bacteria and present in Gram-Negative bacteria. This major difference between gram-positive and Gram-Negative cell wall composition is very critical for health practitioners. 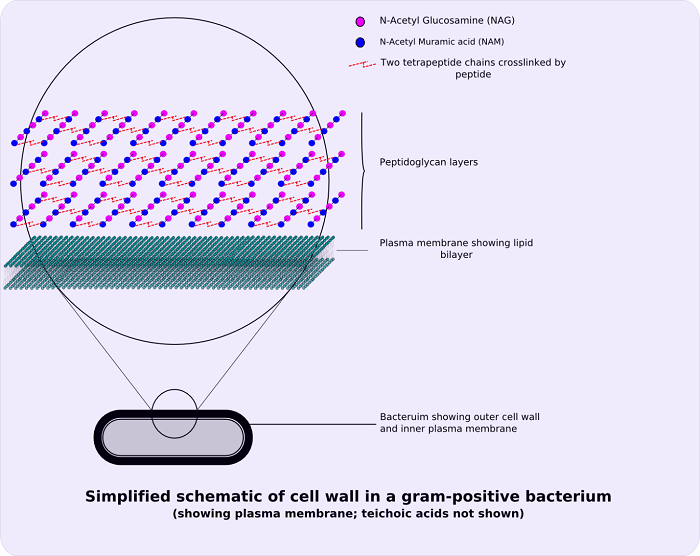
In 1884, Hans Christian Gram invented the staining procedure. The crystal violet dye used in the staining process is retained by the thick peptidoglycan cell wall present in gram-positive bacteria. When seen under a microscope, this process turns gram-positive organisms blue. The color distinguishes gram-positive bacteria they produce when stained. Despite having an outer membrane, gram-negative organisms have a weaker peptidoglycan layer that does not contain the blue dye employed in the first dying phase. The morphology of bacteria is also utilized to distinguish them. Cocci, bacilli, and branching filaments are examples of Gram-positive bacteria. EtiologyStaphylococcus (catalase-positive), which forms clusters, and Streptococcus (catalase-negative), which forms chains, are two Gram-positive cocci. Staphylococci are divided by coagulase-positive (S. aureus) and coagulase-negative (S. epidermidis and S. saprophyticus) staphylococci. Strep. pyogenes (Group A), Strep. agalactiae (Group B), enterococci (Group D), Strep viridans, and Strep pneumonia are several types of streptococcus bacteria. Gram-positive bacilli (rods) are divided into groups based on their spore-producing abilities. Listeria and Corynebacterium are spore-forming rods, but Bacillus and Clostridia are not. Nocardia and actinomyces are also included in the branching filament rods. Spore-forming rods may live in the environment for many years and generate spores. While comparing Gram-Negative and Gram-Positive Bacteria, as already stated, Gram-Positive Bacteria have a thicker peptidoglycan cell wall. The thickness of the peptidoglycan layer in the Gram-Negative cell wall is around 2 to 3 nanometers. Still, it is also covered by an Outer lipid by layer membrane, which is a 20 to 80nm thick polymer. CharacteristicsGram-positive bacteria have the following properties in general:
Only a few species contain a capsule, which is often made up of polysaccharides. Only a few species have flagella, and when they do, they only have two basal body rings to maintain them, while gram-positive bacteria have four. An S-layer is found on the surface of both gram-positive and gram-negative bacteria. The S-layer is connected to the peptidoglycan layer of gram-positive bacteria. The S-layer of Gram-negative bacteria is directly connected to the outer membrane. The presence of teichoic acids in the cell wall is unique to gram-positive bacteria. Lipoteichoic acids, for example, contain a lipid component in the cell membrane that may help bind the peptidoglycan. ClassificationGram staining, together with cell shape, is a quick way to distinguish bacterial species. This staining, growth requirements and antibiotic susceptibility testing, and other morphological and physiologic tests constitute the foundation for bacterial categorization and subdivision. 
Gram-Positive Bacteria originates from a single ancestor and forms a monophyletic group. This is a matter of question for many scientists because of the work done by Carl Woese based on 16S ribosomal RNA phylogenetic analyses along with collaborators and colleagues at the University of Illinois. Woese identified twelve bacterial phyla based on molecular investigations of 16S sequences. Two of them were gram-positive, and their DNA was split according to the amount of guanine and cytosine. The Actinomycetota belonged to the high G + C phylum, whereas the Firmicutes belonged to the low G + C phylum. The Bacillota (low G + C) has a GC concentration of45-60%, which is lower than that of the Actinomycetota. Corynebacterium, Mycobacterium, Nocardia, and Streptomyces are all members of the Actinomycetota. The Outer Cell Membrane's Role in Bacterial TaxonomyThrough the period of time, although the bacteria has always been differentiated on the basis of the Gram staining procedure, that is, the stain retention property, this classification is rather ambiguous. The classification refers to 3 very different aspects of bacteria: the staining result, the envelope organization, and the taxonomic group, which does not necessarily coalesce some bacterial species.A varThe staining procedure has resulted in the formation of the two diverse groups of bacteria, that is, gram-negative and gram-positive. But this classification is quite unreliable because these two groups of bacteria do not form phylogenetic groups. But instead, they form a monophyletic group which implies that they're not as diverse as they should be. 
Gram-positive bacteria are all separated by a single-unit lipid membrane, which has a thick coating (20-80 nm) of peptidoglycan that keeps the Gram stain in place. A single membrane separates a variety of bacteria, but stain gram-negative due to the absence of the peptidoglycan layer, as in mycoplasmas, or their failure to maintain the Gram stain due to cell wall composition, have a close connection to Gram-positive bacteria. Monoderm bacteria is a word used to describe bacterial cells that are separated by a single cell membrane. Gram-negative bacteria have an outer cell membrane that surrounds the bacteria's plasma membrane or cytoplasmic membrane, along with a thin peptidoglycan layer between them. The periplasmic space, also known as the periplasmic compartment, is defined by the existence of inner and outer cell membranes in these cells. Diderm bacteria is the name given to these bacteria. Conserved signature indels in a number of key proteins reinforce the differentiation between monoderm and diderm bacteria (viz. DnaK, GroEL). Monoderms seem to be the more primitive of the two structurally dissimilar types of bacteria. 
From the work that has been done, it is assumed that the outer cell membrane of the gram-negative bacteria has evolved as a protective covering to protect bacteria from any antibiotic that affects the cell membrane and enhance it to act as a mechanism of protection against antibiotic selection pressure. Another observation made in this regard is that Gram-Positive Bacteria are the major producers of antibiotics, and Gram-Negative bacteria are resistant to their counterparts. There are some bacteria that exist as the intermediate form between gram-positive and Gram-Negative bacteria, for example, Deinococcus because they have a thick peptidoglycan layer as well as the outer cell membrane Simple diderms without lipopolysaccharide, archetypical diderm bacteria with lipopolysaccharide in the outer cell membrane, and diderm bacteria with mycolic acid in the outer cell membrane are the three types of diderm bacteria. ExceptionsGram-Positive Bacteria are called monoderms because they have a single lipid bilayer in their cell membrane, whereas Gram-Negative bacteria are called Diderms because of the presence of two lipid bilayers. There are also some gram bacteria that lack a peptidoglycan layer altogether, and hence they are called a gram variable. 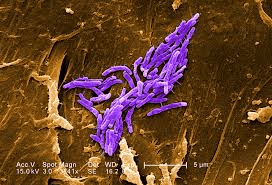
This isn't always the case, as the Deinococcus-Thermus bacteria exhibit gram-positive stains despite having two layers and being structurally similar to gram-negative bacteria. Other exceptions include Chloroflexi, which has only a single layer of a pride o glycan but still, it stains negative. Some Firmicute species don't have gram-positive bacteria. These include members of the Mollicutes class (also known as Tenericutes), which lack peptidoglycan and are gram-indeterminate, and the Negativicutes class, which contains Selenomonas and is gram-negative. PathogenesisSix gram-positive genera are frequently pathogenic in humans in the traditional sense. Streptococcus and Staphylococcus are two types of cocci (sphere-shaped). The remaining organisms are bacilli (rod-shaped bacteria) that may be separated into groups depending on their capacity to produce spores. Corynebacterium and Listeria (a coccobacillus) do not create spores, although Bacillus and Clostridium do. Bacillus is a facultative anaerobe, whereas Clostridium is an obligatory anaerobe. Hence the spore-forming bacteria may be split again depending on their respiration. In addition, three gram-positive taxa that cause plant disease include Rathybacter, Leifsonia, and Clavibacter. Gram-positive bacteria may infect newborn newborns and cause severe and sometimes deadly illnesses. Catabacter hongkongensis, an emerging pathogen belonging to the Firmicutes family, is another novel therapeutically relevant gram-positive bacteria species. 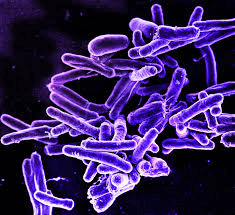
Bacterial metamorphosis is a term used to describe the transformation of bacteria. Transformation is the process of transferring genetic material from one bacterial cell to another bacterial cell. Bacteria have devised many different methods for the process of transformation. That transformation is divided into two broad categories; the first one is conjugation, in which the genetic material is transferred with the help of a pilus that connects two bacterial cells. Another method is transduction, in which the bacteria uses a bacteriophage virus to inject the bacterial donor DNA into the recipient host bacteria. In transduction, the exchange of genetic material occurs with the help of a carrier. This technique is also called horizontal gene transfer because it is not the transfer of genetic material across the generation through the process of sexual reproduction, but instead, they are transferred between any two species of bacteria. The genetic material travels through the intermediary medium during transformation, and absorption is entirely reliant on the recipient bacteria. About 80 kinds of bacteria were known to be capable of transformation as of 2014, about equally split between gram-positive and gram-negative bacteria; however, the number may be an exaggeration since some of the findings are based on single studies. Transformation of medically significant bacteria, including Streptococcus pneumoniae, Streptococcus mutans, Staphylococcus aureus, and Streptococcus sanguinis, as well as gram-positive soil bacteria like Bacillus subtilis and Bacillus cereus, has been researched. EpidemiologyAccording to the SCOPE project, the data is surveillance and control of pathogens of epidemiologic importance. In only two decades, rates of bloodstream infection mortality have risen by 78 percent. Gram-positive bacteria exhibit a wide range of growth and resistance patterns. There have been records of infections caused by Gram-Positive Bacteria. In 1995, these bacteria were responsible for 62% of all the bloodstream infections, and in 2000, the infections increased to 76%. On the other hand, the infection rate of Gram-Negative Bacteria was much less, 22% in 1995 and 14% in 2000. PathophysiologyStaphylococcus aureus is a gram-positive, catalase-positive, and coagulase-positive cocci that grow in clusters. S. aureus is responsible for a number of inflammatory disorders like skin infections, osteomyelitis, pneumonia, endocarditis, and septic arthritis. The other problems and diseases caused by this bacteria include toxic shock syndrome, food poisoning, and scalded skin syndrome, also called an exfoliative toxin. Staphylococcus epidermidis is a novobiocin-sensitive gram-positive, catalase-positive, coagulase-negative cocci in clusters. Whenever staphylococcus epidermidis infects any prosthetic device and IV catheters, it results in the formation of biofilms. Staphylococcus saprophyticus is a typical flora of the vaginal tract and perineum that is novobiocin resistant. The bacteria are also responsible for causing the most prevalent urinary tract infection. Another Gram-Positive Bacteria is staphylococcus pneumoniae which is responsible for causing otitis media, meningitis, sinusitis, and pneumonia. It is a lancet-shaped encapsulated diplococcus bacterium. Streptococcus viridans, which includes Streptococcus mutans and Streptococcus mitis, is a genus of bacteria present in the natural flora of the oropharynx that causes dental caries and subacute bacterial endocarditis (Strep. sanguinis). The gram-positive group, A cocci Streptococcus pyogenes, may cause pyogenic infections (pharyngitis, cellulitis, impetigo, erysipelas), toxigenic infections (scarlet fever, necrotizing fasciitis), and immunologic infections (scarlet fever, necrotizing fasciitis) (glomerulonephritis and rheumatic fever). S. pyogenes infections are detected using the ASO titer. A bacterium that colonizes the vaginal canal and is most prevalent in newborn babies is Streptococcus agalactiae. It is a gram-positive group B coccus. Enterococci are gram-positive group D cocci that may cause biliary tract infections and urinary tract infections. They are mostly present in the intestinal flora. VRE (vancomycin-resistant enterococci) are a common source of nosocomial infections in hospitals. 
Gram-Positive RodsClostridia tetani, C. botulinum, C. perfringens, and C. difficile are gram-positive spore-forming rods. Antibiotic usage (clindamycin/ampicillin), PPI use, and recent hospitalization are all common causes of C. difficile. The most common treatment is oral vancomycin. Bacteria that are obtained from reheated rice spores or undercooked food is Bacillus cereus which is responsible for diseases like non-bloody diarrhea, vomiting, and nausea. Bacillus cereus is a Gram-Positive Bacteria under the category of rod-shaped bacteria. Another Gram-Positive Bacteria, which is also a spore-forming rod, is anthracis, and it is responsible for ulcers. The gram-positive club-shaped rod Corynebacterium diphtheria may cause pseudomembranous pharyngitis, myocarditis, and arrhythmias. Diphtheria is prevented with toxoid vaccinations. Listeria monocytogenes is a gram-positive rod that may be contracted by consuming cold deli meats and unpasteurized dairy products and through vaginal transmission after childbirth. Listeria may cause newborn meningitis, immunocompromised meningitis, gastroenteritis, and septicemia in those who are immunocompromised. Ampicillin is used in the treatment. Physical and Historical ContextIt's critical to recognize sepsis patients and get the requisite blood cultures and lab tests done. Physical signs and symptoms include • Bullous impetigo • Draining sinus tracts • Erythema • Fever • Murmur if endocarditis is present • Petechiae if toxic shock syndrome is present • Superficial abscesses. EvaluationThese laboratory tests are relevant after a gram-positive organism infection is suspected: Complete blood count (CBC); electrolytes; blood cultures; procalcitonin level; echocardiogram if endocarditis is suspected; joint aspiration if a septic joint is suspected Management / TreatmentAlexander Fleming introduced penicillin as the first antibiotic during World War II in 1928. Staph and Enterococcus are not covered by penicillin, which is mostly used to treat streptococcal infections. Staph (MSSA) and Strep are penicillinase-resistant organisms (nafcillin, oxacillin, cloxacillin, dicloxacillin). Piperacillin and ticarcillin are anti-pseudomonal penicillins that work against gram-positive, gram-negative, pseudomonas, and anaerobes. Gram-positives, gram-negatives, and anaerobes are all covered by carbapenems. Oral medicines such as trimethoprim/sulfamethoxazole, clindamycin, and doxycycline are used to treat mild to severe MRSA infections. It's worth noting that trimethoprim/sulfamethoxazole raises warfarin levels, resulting in a higher INR. MRSA acquired in the community and in hospitals is treated with vancomycin, linezolid, daptomycin, and tigecycline. Renal dosage with trough values of 15 to 20 is required for vancomycin. If a patient is allergic to vancomycin, linezolid is an alternative. CBC should be monitored regularly to minimize bone marrow suppression, neutropenia, thrombocytopenia, and anemia. Vancomycin-resistant enterococci may be treated with linezolid, daptomycin, or tigecycline. Differential Diagnosis
Prognosis after infection with gram-positive organisms, the prognosis varies. Elderly people have the greatest death rates because their immune systems are weakened, and they have a less physiologic reserve. Improving the Performance of Healthcare TeamsTo appropriately treat patients with chosen antibiotics, health professionals, such as physicians, nurses, and pharmacists, must be aware of risk factors. Pharmacists must carefully monitor vancomycin trough levels to reduce mortality in Staph aureus patients. They must also check medications for dosage and interactions and educate patients on the need to complete all antibiotics given. Infection control nurses assess nosocomial illnesses and put in suitable place rules. The greatest results will come from an interprofessional approach. 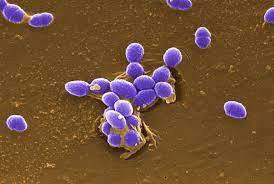
Outcomes:Infection management is improved by screening for methicillin-resistant Staphylococcus aureus (MRSA) risk factors. All MRSA risk factors are patients over the age of 65, urinary catheters, prior antibiotic therapy within the preceding three months, trauma, and those admitted from a long-term hospital.
Next TopicBacterial Morphology
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share









