What is a nucleotideNucleotide is an organic molecule. It is the building block of DNA and RNA and is made of three components that include a phosphate group, a 5-carbon sugar, and a nitrogenous base. The nitrogenous base can be of four types or we can say that there are four different nitrogenous bases in DNA that are adenine, cytosine, guanine and thymine. However, in RNA instead of thymine, uracil is present, in other words, thymine is replaced by uracil. So, it is the nucleotides that make the DNA (genetic material) of all living organisms. Structure of Nucleotide:A nucleotide comprises three units that are linked through covalent bonds.
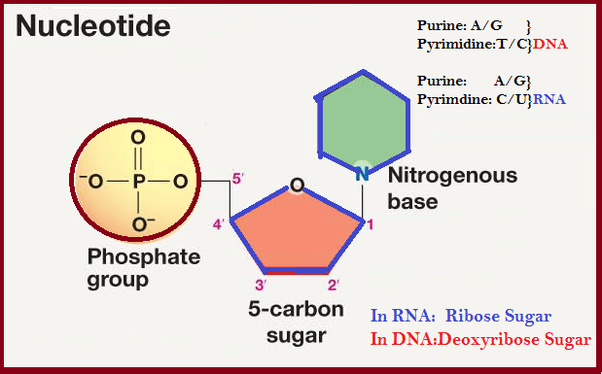
1) Nitrogenous bases or Nucleotide Bases:A nucleotide is defined or identified by its chemical base. There are a total of five chemical bases:
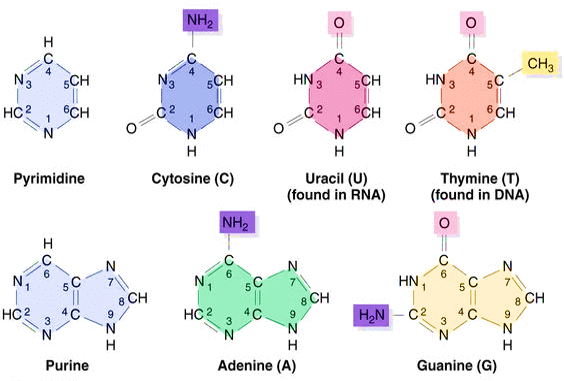
A nucleotide is named on the basis of the type of base and phosphate residue. For example, an Adenine nucleotide that has one phosphate group is known as adenosine monophosphate. Adenosine is derived from the Adenine base of the nucleotide and monophosphate indicates that it has one phosphate group. A base is represented by a letter and it can be either purine or pyrimidine as described below: Adenine (A): This base is a purine, which is a family of nitrogenous bases. Its chemical formula is C5H5N5 and it has a double-ringed structure like other purines. A nucleotide with adenine as a base is called Adenine. It has two hydrogen bonds that help stabilize the structure of nucleic acids such as DNA, RNA. In DNA, adenine is bonded with thymine and thus it makes pair with thymine. Whereas, in RNA, it is bonded with uracil. Cytosine (C): This base is a pyrimidine which is a molecule whose chemical formula is C4H5N3O. A cytosine based nucleotide is known as cytosine. Its structure has only one ring and it makes a pair with guanine in both nucleic acids, DNA and RNA. However, when it is free it may act as a co-enzyme, which helps convert ADP to ATP. Guanine (G): This base is also a purine, a molecule with a double ring structure and chemical formula C5H5N5O. A nucleotide that carries guanine as a base is called guanosine. It bonds with cytosine through three hydrogen bonds in both nucleic acids DNA and RNA. Due to the three hydrogen bonds, it is slightly stronger than the adenine-thymine bond that has only two hydrogen bonds. Thymine (T): It is pyrimidine base with chemical formula C5H6N2O2. A nucleotide based on the thymine is called thymine. It pairs or bonds with adenine in nucleic acids that assist in stabilizing the nucleic acid structures. Uracil (U): Uracil is a pyrimidine base with chemical formula C4H4N2O2. A uracil-based nucleotide is called uracil. Uracil is a demethylated form of thymine and is present in RNA in place of thymine. Demethylation is a chemical process in which a CH3 (methyl group) is removed from a molecule. These bases can combine with sugars and phosphates to form free nucleotides and can bond with each another to form nucleic acids like DNA and RNA. 2) Pentose Sugars or Pentose Monosaccharides:It is one of the three components of a nucleotide that is needed to make this molecule. It is five-carbohydrate sugar. A nucleotide can have one of the following two sugars:
So, there can be two types of pentose sugars in nucleotides; ribose (C5H10O5) and deoxyribose (C5H10O4). The ribose sugar has a -OH group, whereas, the deoxyribose sugar has -H at C-2 position. The nucleotides that contain ribose are called ribonucleotides or ribotides and are found only in RNA, whereas, nucleotides that contain deoxyribose are called deoxyribonucleotides or dexoyribotides. DNA is a nucleic acid with a deoxyribose sugar so it is named deoxyribonucleic acid. Similarly, RNA has ribose sugar so it is named ribonucleic acid. A nitrogenous base which is attached to a sugar molecule is called Nucleoside. 3) Phosphate Groups:A nucleotide can have one, two or three phosphate groups. Accordingly, they are known as monophosphate (with one phosphate group), diphosphates (with two phosphate groups) and triphosphates with three phosphate groups. Phosphate is attached to the sugar of nucleoside by an ester bond with the 5-Carbon hydroxyl group. Thus, a nucleotide is formed from nucleoside. So, a nucleotide is required to have at least one phosphate group. The phosphate group of a nucleotide is attached to the 3rd C-OH group of the sugar of the other nucleotide and thus forms 5' - 3' linkage. The antiparallel strands of nucleotides in DNA are linked with hydrogen bonds between nitrogenous bases. Purines bases pairs with pyrimidine bases. For example, A pairs or attached with T by two H bonds, and G pairs or is bonded with C through three H bonds. However, in RNA (Ribonucleic Acid), A pairs with U (Uracil) instead of Thymine. Functions of Nucleotides:
Formation of Nucleosides and Nucleotides: 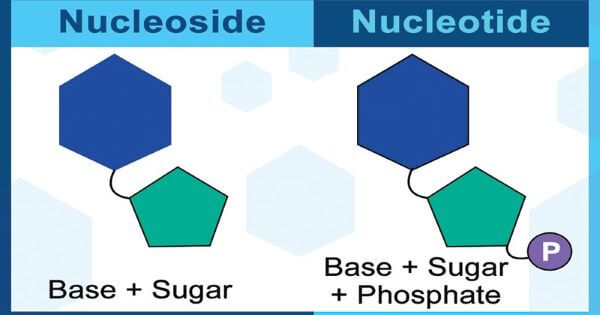
Formation of Nucleosides:A nucleoside is formed when a pentose sugar combines with a nitrogenous base through N-glycosidic base. 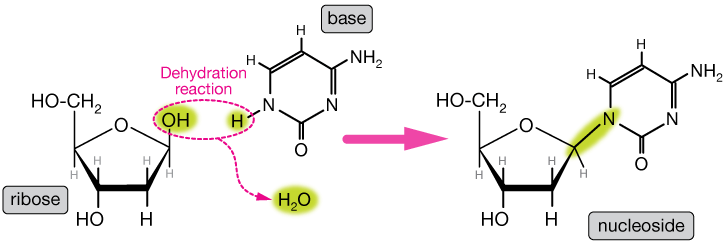
The C-1 carbon of pentose sugar is attached to N-9 of a purine or N-1 of a pyrimidine to form a nucleoside. A nucleoside that has ribose sugar is called ribonucleoside or riboside and a nucleoside that has deoxyribose sugar is called deoxyribonucleoside or deoxyriboside. Some examples of ribonucleoside include adenosine (adenine + ribose), guanosine (guanine + ribose), and thymidine (thymine + ribose) and cytidine (cytosine + ribose). Similarly, some of the corresponding examples of deoxyribonucleosides include deoxyadenosine, deoxyguanosine, deoxythymidine, deoxycytidine, etc. Formation of nucleotides I How is a nucleotide formed?Nucleotides are formed from nucleoside. They are phosphate esters of nucleosides. A phosphate is bonded with 5' carbon of sugar of a nucleoside to form a nucleotide or nucleoside monophosphate. Some examples of nucleotides include adenosine monophosphate (AMP), Deoxyadenosine monophosphate (dAMP), Guanosine monophosphate (GMP), Deoxyguanosine monophosphate (dGMP). These nucleotides have one phosphate. The nucleotides that have more than one phosphate group are called higher nucleotides such as Adenosine Triphosphate (ATP), Adenosine Diphosphate (ADP), Guanosine Triphosphate (GTP), etc. So, higher nucleotides are actually nucleoside diphosphates and nucleoside triphosphates. The higher nucleotides occur in a free state and they carry high energy bonds called phosphoanhydride bonds that exist between phosphates. ATP is the most common energy molecule in the cell that releases the required energy when it breaks down to form ADP with the release of energy. The hydrolysis of ATP releases lots of free energy that is required to carry out cellular chemical reactions, e.g. ATP + H2O -- > ADP + Pi + Energy
Next TopicChromosome
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share









