Apoptosis DefinitionApoptosis is a type of programmed cell death that occurs in multicellular organisms. It is a highly regulated process that allows cells to self-destruct in a controlled and orderly manner, without causing damage to surrounding tissues. As it enables the removal of surplus or damaged cells, apoptosis is essential for the growth and upkeep of healthy tissues. This process is characterized by a series of biochemical and molecular events that result in the dismantling of the cell, including the condensation of chromatin, the fragmentation of DNA, the cleavage of cytoskeletal and nuclear proteins, and the formation of apoptotic bodies. 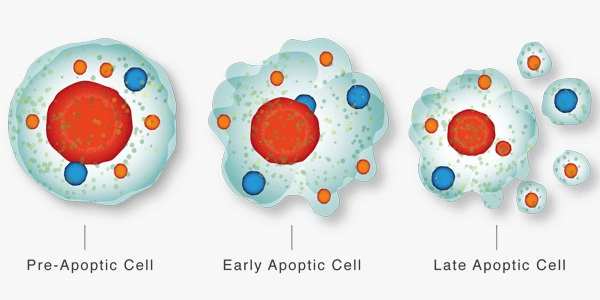
Numerous biological processes, including tissue homeostasis, the immune response, and embryonic development, involve apoptosis. It also serves as a defense mechanism against cancer, as it eliminates cells with DNA damage or mutations that could potentially lead to tumor formation. Apoptosis dysregulation has been linked to a number of human illnesses, including cancer, immunological disorders, neurodegenerative diseases, and developmental abnormalities. Understanding the mechanisms of apoptosis and how it is regulated may lead to the development of new treatments for these diseases. Historical Background of ApoptosisThe concept of programmed cell death was first proposed in the early 20th century by the German biologist Carl Vogt, who suggested that the death of cells in the developing embryo was a normal and necessary process. However, the term "apoptosis" was not coined until 1972. It was introduced by the Australian pathologist John Kerr, who observed a distinct type of cell death characterized by cell shrinkage and fragmentation in tissue samples. In the following years, other researchers such as Andrew Wyllie and Alastair Currie further investigated this phenomenon and described the characteristic morphological changes that occur during apoptosis, including chromatin condensation and the formation of apoptotic bodies. In the 1980s and 1990s, the molecular mechanisms of apoptosis began to be elucidated, with the discovery of key genes such as BCL-2 and CED-9 that regulate the process. The identification of caspases, a family of proteases that are activated during apoptosis, also provided further insight into the molecular events that occur during cell death. Today, apoptosis is recognized as a fundamental process in biology, with important implications for both normal development and disease pathology. The historical background of apoptosis reflects a long and ongoing process of scientific discovery, and the continuing study of this process promises to yield important insights into the workings of the cell and the mechanisms that regulate cell fate. Apoptosis and Programmed Cell DeathApoptosis is a type of programmed cell death, which is a natural and necessary process that occurs during the development and maintenance of healthy tissues. Programmed cell death is an umbrella term that encompasses various types of cell death that are controlled by specific molecular pathways. Unlike necrosis, which is a form of uncontrolled cell death that results from external damage or injury, programmed cell death is an active process that is regulated by the cell's own machinery. Apoptosis is one of the most well-studied types of programmed cell death and is characterized by a series of morphological and biochemical changes that result in the controlled dismantling of the cell. Apoptosis plays a critical role in a wide range of biological processes, including embryonic development, tissue homeostasis, and the immune response. It is also involved in the elimination of damaged or abnormal cells, such as those with DNA damage or mutations that could potentially lead to tumour formation. Other types of programmed cell death include autophagy, which is the process of self-digestion that occurs when cells are deprived of nutrients, and necroptosis, which is a programmed form of necrosis that occurs in response to certain stimuli such as viral infections or inflammatory signals. The study of programmed cell death has important implications for understanding the mechanisms that regulate cell fate and tissue homeostasis, as well as for the development of new therapies for diseases such as cancer and neurodegenerative disorders. Apoptosis and CancerApoptosis plays a critical role in preventing the development of cancer by eliminating cells with DNA damage or mutations that could potentially lead to tumor formation. When apoptosis is dysregulated, such as when the pro-apoptotic pathways are disrupted or anti-apoptotic factors are upregulated, it can contribute to the development and progression of cancer. In many types of cancer, there is a decrease in the ability of cells to undergo apoptosis, which allows cancer cells to evade normal cell death and continue to proliferate. Cancer cells may also acquire mutations in the genes that regulate apoptosis, leading to an imbalance in the pro- and anti-apoptotic pathways. In addition, cancer cells may secrete factors that suppress apoptosis in neighboring cells or activate signaling pathways that promote cell survival. This can create a microenvironment that is permissive for cancer growth and metastasis. Therapies that target the apoptotic pathway have shown promise in the treatment of cancer. For example, some chemotherapeutic drugs work by inducing apoptosis in cancer cells, while others target specific proteins involved in apoptosis regulation. Understanding the role of apoptosis in cancer development and progression may lead to the development of new therapies that selectively target cancer cells while preserving healthy tissues, and ultimately improving the prognosis for cancer patients. Apoptosis and Neurodegenerative DiseasesApoptosis has been implicated in the pathogenesis of neurodegenerative diseases, which are a group of disorders characterized by progressive loss of function and death of nerve cells in the brain or spinal cord. Dysregulation of apoptosis in the central nervous system (CNS) can contribute to the accumulation of abnormal proteins, inflammation, and neurotoxicity, which are hallmarks of many neurodegenerative diseases. There is evidence that apoptosis is elevated in damaged brain areas of various neurodegenerative disorders, including Alzheimer's disease & Parkinson's disease. For instance, in Alzheimer's disease, amyloid beta protein buildup in the brain is considered to cause nerve cells to undergo apoptosis, resulting in neurodegeneration. Similarly, in Parkinson's disease, the accumulation of misfolded alpha-synuclein protein in neurons can induce apoptosis and contribute to the death of dopaminergic neurons in the substantia nigra region of the brain. On the other hand, in other neurodegenerative diseases such as Huntington's disease, there is evidence that a failure of apoptosis may be implicated. In Huntington's disease, mutant huntingtin protein accumulates in neurons, leading to impaired clearance of damaged proteins and disruption of cellular processes, including apoptosis. The role of apoptosis in neurodegenerative diseases is complex and multifactorial, involving a variety of molecular pathways and cellular interactions. The balance between pro-apoptotic and anti-apoptotic factors in the CNS can determine the fate of neurons and contribute to disease progression. Understanding the role of apoptosis in neurodegenerative diseases may lead to the development of novel therapeutic strategies aimed at modulating apoptotic pathways to protect nerve cells from death and potentially slow or halt disease progression. However, further research is needed to elucidate the precise mechanisms involved and to develop effective therapies for these complex and devastating diseases. Mechanisms of Apoptosis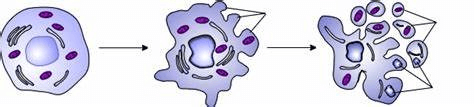
Apoptosis is a tightly controlled process that involves a number of molecular activities, such as the activation of certain signalling pathways, modifications to gene expression, and changes to the appearance and function of cells. Extrinsic (or death receptor-mediated) & intrinsic (or mitochondrial-mediated) pathways are the two basic types of apoptosis processes. Apoptosis pathways and signalling involve a complex interplay of multiple proteins and molecules that regulate the initiation, progression, and execution of programmed cell death. The two main pathways of apoptosis, the extrinsic and intrinsic pathways, are regulated by distinct sets of proteins and signalling molecules. Extrinsic PathwaySpecific ligands that bind to their corresponding cell surface receptors, also known as death receptors, such as Fas ligand or tumour necrosis factor (TNF), start the extrinsic pathway. The binding of these ligands to death receptors triggers the formation of a death-inducing signalling complex (DISC) that activates caspase-8, a protease that initiates a cascade of downstream events that ultimately result in cell death. The DISC complex contains multiple proteins, including the adapter protein Fas-associated death domain (FADD), the pro-caspase-8 enzyme, and other signalling proteins. When ligand binding occurs, death receptors aggregate and FADD binds to the receptor's intracellular death domain, recruiting and activating pro-caspase-8. After becoming active, caspase-8 cleaves & activates downstream caspases, which causes apoptosis to occur. Intrinsic PathwayThe intrinsic pathway is triggered by a number of cellular stressors, such as DNA damage, oxidative stress, or nutritional deficiency. The B-cell lymphoma 2 (BCL-2) family of proteins, which are found on the outer membrane of mitochondria, balance pro- and anti-apoptotic proteins to control this process. Pro-apoptotic BCL-2 family members like Bax and Bak encourage the release of cytochrome c from the mitochondria when cells are subjected to apoptotic stimuli. This activates caspase-9, which sets off a series of subsequent events that eventually lead to cell death. BCL-2 and BCL-xL are anti-apoptotic BCL-2 family members that help cells survive by preventing the release of cytochrome c from the mitochondria. Both the extrinsic and intrinsic pathways converge on the activation of caspases, a family of cysteine proteases that are responsible for the execution of apoptotic cell death. Caspases cleave specific substrates, including cytoskeletal proteins and DNA repair enzymes, which ultimately lead to the characteristic morphological changes of apoptosis, such as cell shrinkage, nuclear condensation, and fragmentation. Overall, the mechanisms of apoptosis are complex and involve a wide range of molecular interactions and cellular processes. For tissues to remain in a state of homeostasis and to avoid illnesses like cancer and neurological disorders from emerging, apoptotic pathways must be properly regulated. Other Signaling PathwaysSeveral other signaling pathways can modulate apoptosis, including the PI3K/AKT, MAPK/ERK, and NF-?B pathways. These pathways can regulate the expression of pro- and anti-apoptotic proteins and affect the balance between cell survival and death signals. Detection of ApoptosisSeveral methods are used to detect and measure apoptosis in cells and tissues. Some commonly used methods include the following:
Future Directions in Apoptosis ResearchApoptosis research has advanced significantly over the past few decades, providing insight into the molecular mechanisms of programmed cell death and its implications in various diseases. However, there are still several areas of research that require further investigation. Some of the future directions in apoptosis research include the following:
The Bottom LineIn conclusion, apoptosis is a highly regulated form of programmed cell death that plays a crucial role in various physiological and pathological processes. It is a complex and dynamic process involving a series of molecular events, including caspase activation, mitochondrial outer membrane permeabilization, and DNA fragmentation. Apoptosis is essential for normal development, tissue homeostasis and immune response, but dysregulation of apoptosis can lead to many diseases, including cancer, neurodegenerative diseases, autoimmune diseases and others.
Next TopicDivision Definition
|
 For Videos Join Our Youtube Channel: Join Now
For Videos Join Our Youtube Channel: Join Now
Feedback
- Send your Feedback to [email protected]
Help Others, Please Share










